Sign up for our newsletter!
Your data will be handled in compliance with our privacy policy.
Your data will be handled in compliance with our privacy policy.

E-fuel is an umbrella term for fossil-free alternatives to gasoline, diesel, and other fossil fuels. These fuels are produced by converting fossil-free hydrogen and capturing carbon dioxide. They can be used in existing engines without increasing the amount of carbon dioxide (CO₂) in the atmosphere. Is this the silver bullet that allows us to drive cars and other vehicles with a clear conscience?
Thomas Barregren • January 31, 2024
It’s evident to everyone that we can’t keep pumping oil and gas to fuel our vehicles for much longer; every liter increases the amount of carbon dioxide in the atmosphere and warms the Earth. But that doesn’t mean we must scrap or rebuild all vehicles. With hydrogen produced by electrolyzers powered with fossil-free electricity and captured carbon dioxide, it is possible to create carbon-neutral equivalents to today’s fuel. These are called electrofuels, or e‑fuels, and you can learn more about them in this post.
Virtually all motor vehicles – cars, trains, boats, planes, and rockets – are powered by mixtures of hydrocarbons. These chemical compounds are chains and rings of different numbers of carbon atoms from which hydrogen atoms dangle like charms.
The hydrocarbons we use to power vehicles come from fossils, mainly algae and plankton, which have been transformed over hundreds of thousands of years into gases and the viscous yellow-black liquid we call petroleum, crude oil, or simply oil. These natural resources are refined, cracked, and blended into liquid natural gas (LNG), liquid petroleum gas (LPG), methanol, jet fuel, gasoline, kerosene, diesel, or other petroleum products we use to fuel our vehicles.
As you know, we are extracting more gas and oil than nature can restore. This is not sustainable, which the 1973 oil crisis made people painfully aware of. But this is a minor concern compared to the climate crisis we are heading straight into.
Burning fossil fuels releases a lot of carbon dioxide (CO2) into the atmosphere. This gas traps heat from the sun, like a seal, causing the Earth’s temperature to rise similar to how it does in a greenhouse. This has been known since 1896.
Yes, that’s right. Humanity has known for over 120 years that our appetite for fossil fuels will create the greenhouse effect. Yet we went for fossil fuel vehicles instead of continuing the early development of alternatives such as electric cars and synthetic fuels.
Only under the increasingly imminent threat of melting ice, overflowing seas, water shortages, and barren farmland have we begun to look at alternatives. Many ideas from the late 19th and early 20th centuries are experiencing a renaissance. Most apparent is the resurrection of the battery electric vehicle (BEV).
But that’s not the only option. Other old ideas that have been revived are using hydrogen as fuel, either directly or via conversion to electricity. But perhaps the most promising short-term solution is synthetic fuel made from green hydrogen. So, let’s take a closer look at it.
Electrofuel, or shorter e‑fuel, is an umbrella term for e‑LNG, e‑LPG, e‑methanol, e‑jet fuel, e‑gasoline, e‑kerosene, e‑diesel and other synthetic fuels that can replace their non-prefixed counterparts without any modification to the engines that use them.
E‑fuels are produced from carbon dioxide (CO2), captured from the atmosphere, and hydrogen, produced by water electrolysis using fossil-free electricity (green hydrogen or pink hydrogen).
The first synthetic fuel was produced in 1920s Germany by Franz Fischer and Hans Tropsch. Their method has since been refined and further developed. In the early 2000s, synthetic fuel got a renaissance when the idea of e‑fuel began to take shape. Drivers today are car manufacturers like Porsche and Mazda, oil companies like Exxon Mobile, Circle K, Eni, and Repsol, and industrial groups like Siemens, Bosch, Mahle, and ZF.
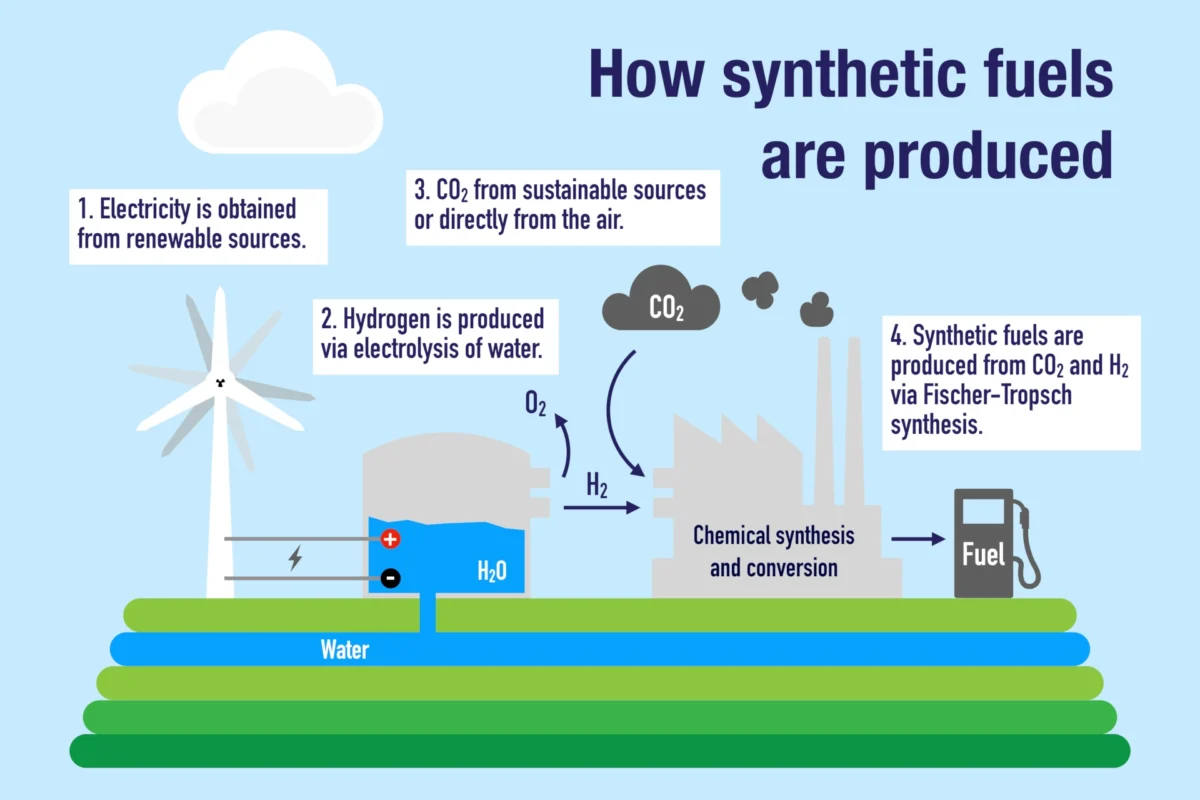
E‑fuels contain essential hydrocarbons found in their conventional counterparts, enabling them to replace these fossil fuels seamlessly.
However, this raises a question: if both e‑fuel and its fossil fuel counterpart emit the same amount of carbon dioxide when burned, what is the benefit?
Sure, e‑fuel releases as much carbon dioxide into the atmosphere when combusted as its fossil-based counterparts. However, this amount is the same as either captured before it got into the atmosphere or taken from the atmosphere during the production. Thus, the net contribution is zero. One could say that e‑fuels recycle carbon dioxide.
So, can we say that synthetic fuel is carbon neutral?
To make such a bold claim, the process and the raw material must be carbon neutral. This means that the production plant must be powered by fossil-free energy. It also rules out black, brown, and gray hydrogen as a feedstock; such hydrogen is produced from coal and natural gas, which produces huge carbon dioxide emissions.
To be truly climate-neutral, green or pink hydrogen must be used. Both are produced by the electrolysis of water. Green hydrogen uses renewable electricity from the sun, wind, or water, while pink hydrogen uses electricity from nuclear power plants.
The prefix ‘electro’ or just ‘e’ refers to hydrogen produced by water electrolysis using fossil-free electricity (green hydrogen or pink hydrogen). The complete production process must run on fossil-free energy sources to earn the prefix.

E‑fuel has three significant benefits in addition to being climate-neutral:
These advantages have given e‑fuel a boost. Around the world, small and large e‑fuel projects are underway.
Regarding e‑fuel production, the Texas-based company HIF Global has come the furthest.
On 20 December 2022, they inaugurated HIF Haru Oni, the first operating e‑fuel facility in the world. The facility, located in Chile, is complete with wind turbines to generate electricity, electrolyzers to convert the electricity into hydrogen, and a direct air capture (DAC) unit to extract carbon dioxide from the atmosphere. The plant produces e‑LPG, e‑methanol, and e‑gasoline. When fully operational, the plant will produce 130,000 liters of e‑fuel annually.
Impressive. But HIF Haru Oni is nothing compared to the company’s next plant. In Texas, they are building the HIF Matagorda eFuels Facility, which, when completed in 2027, will produce 750 million liters of e‑fuel each year. The electrolyzers will have a total expected capacity of about 1.8 gigawatts and produce about 300,000 tonnes of green hydrogen annually.
And it doesn’t stop there. HIF plans to build 12 plants of the same mega-size distributed across Chile, the US, and Australia.
One of the largest investors in the company is the German car company Porsche. They have invested over 100 million USD in e‑fuel. Porsche plans to run its entire Supercup racing series and all the cars at its Porsche Experience Centres on the fuel.

E‑fuel production facilities are also being built in Europe. FlagshipONE, outside Örnsköldsvik in northern Sweden, will produce 55,000 tons of e‑methanol when it is operational in 2025. This makes it the largest of its kind in Europe.
The four electrolyzers used by FlagshipONE will have a total capacity of about 70 megawatts and produce about 12 tons of green hydrogen per year.
The carbon dioxide will be sourced from the adjacent municipal heat and power plant, which burns biomass. The plant will also supply steam and water to the process. The excess heat will then be fed into the municipality’s local district heating network.
FlagshipONE will supply an increasing number of ships that run on methanol.

Naysayers and detractors of e‑fuel claim it is a dead end; e‑fuel is costly and a waste of energy.
They may be right regarding passenger cars and many other types of land transportation. Most of these vehicles are likely to be battery-powered. That technology has come a long way and is much cheaper per kilometer driven than e‑fuel.
But otherwise, they are wrong. Fuel produced synthetically from green hydrogen and captured carbon dioxide will be one of several successful solutions to power vehicles. No single solution works for everyone and everything, so a variety of solutions are needed.
Shipping is a good example. Most experts agree that e‑fuel is the only viable carbon-neutral fuel for large ships like container vessels. These ships use highly polluting fossil fuels, responsible for 3% of the world’s CO2 emissions. We need e‑fuel to address this. Batteries for such ships would be too huge and heavy.
The same goes for airplanes, which also cannot have large and heavy batteries on board.
In addition to these mega markets, there will also be niche markets. One is vehicles that can be used where charging from a reliable, clean electricity grid is impossible.
Another is sports cars, racing cars, and vintage cars, where the sound of an engine revving, the weight, or the preservation of cultural heritage is more important.
In short, the future of e‑fuels is bright and has already begun.
Your data will be handled in compliance with our privacy policy.